AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Formal charge of carbon in h2co12/9/2023 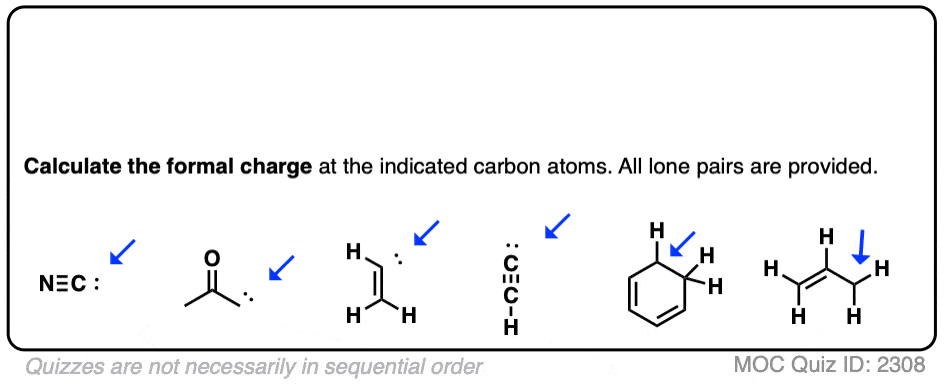
There are several things that should be checked before and after drawing the resonance forms. The reader must know the flow of the electrons. Make sure the arrows are clear including the single and half headed arrow. The tail of the arrow begins at the electron source and the head points to where the electron will be. # of valence electrons- (#non bonding electrons + 1/2 #bonding electrons)Ĭurved arrow notation is used in showing the placement of electrons between atoms. Assigning formal charges to an atom is very useful in resonance forms.įormal charge is calculated using this format: An atom with many electrons will have a negative charge. Atoms that are missing one or more electrons will have a positive charge. This is why formal charges are very important. Remember, the best resonance structure is the one with the least formal charge. These structures used curved arrow notation to show the movement of the electrons in one resonance form to the next.įormal charges are used in Chemistry to determine the location of a charge in a molecule and determine how good of a Lewis structure it will be. They are drawn with a double-headed arrow between them to show the actual structure is somewhere between the resonance structures. 
The more resonance forms a molecule has makes the molecule more stable. Molecular Structure: The structure of a molecule is often dictated by the types and quantities of the elements that it contains. If a compound is organic, carbon is usually the central atom. The better ones have minimal formal charges, negative formal charges are the most electronegative atoms, and bond is maximized in the structure. To calculate the formal charge, take the normal valence of the atom and subtract the number of bonds around the atom. Not all resonance structures are equal there are some that are better than others. Resonance structures are a better depiction of a Lewis dot structure because they clearly show bonding in molecules. Resonance forms differ only in arrangement of electrons. Isomers have different arrangement of both atoms and electrons. 
Resonance structures are not in equilibrium with each other. First resonance structures are not real, they just show possible structures for a compound. There are some basic principle on the resonance theory.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
